Early-stage translational strategy and preclinical decision support.

Pulsus Development is a specialised unit within Pulsus Pharma Consultancy focused
on early-stage asset evaluation and translational strategy.
It supports pharmaceutical and biotech organisations before clinical development is
fully defined, helping determine whether, how, and in whom a therapeutic concept
should progress.
Working with a network of scientific partners, we transform preclinical data into
strategic direction, not just scientific interpretation.
We focus on decision-making under uncertainty, where early evidence must inform high-stakes development choices. Our work includes:
Critical evaluation of preclinical evidence and study design
Mechanism of action validation and translational relevance
Identification of viable clinical hypotheses and target populations
Early definition of development pathways
Integration of preclinical findings with clinical and regulatory context
Our Translational Bridge approach connects early scientific evidence to future clinical and real-world application.
We position preclinical findings within:
This ensures that early-stage decisions are not only scientifically sound, but strategically aligned with downstream success.
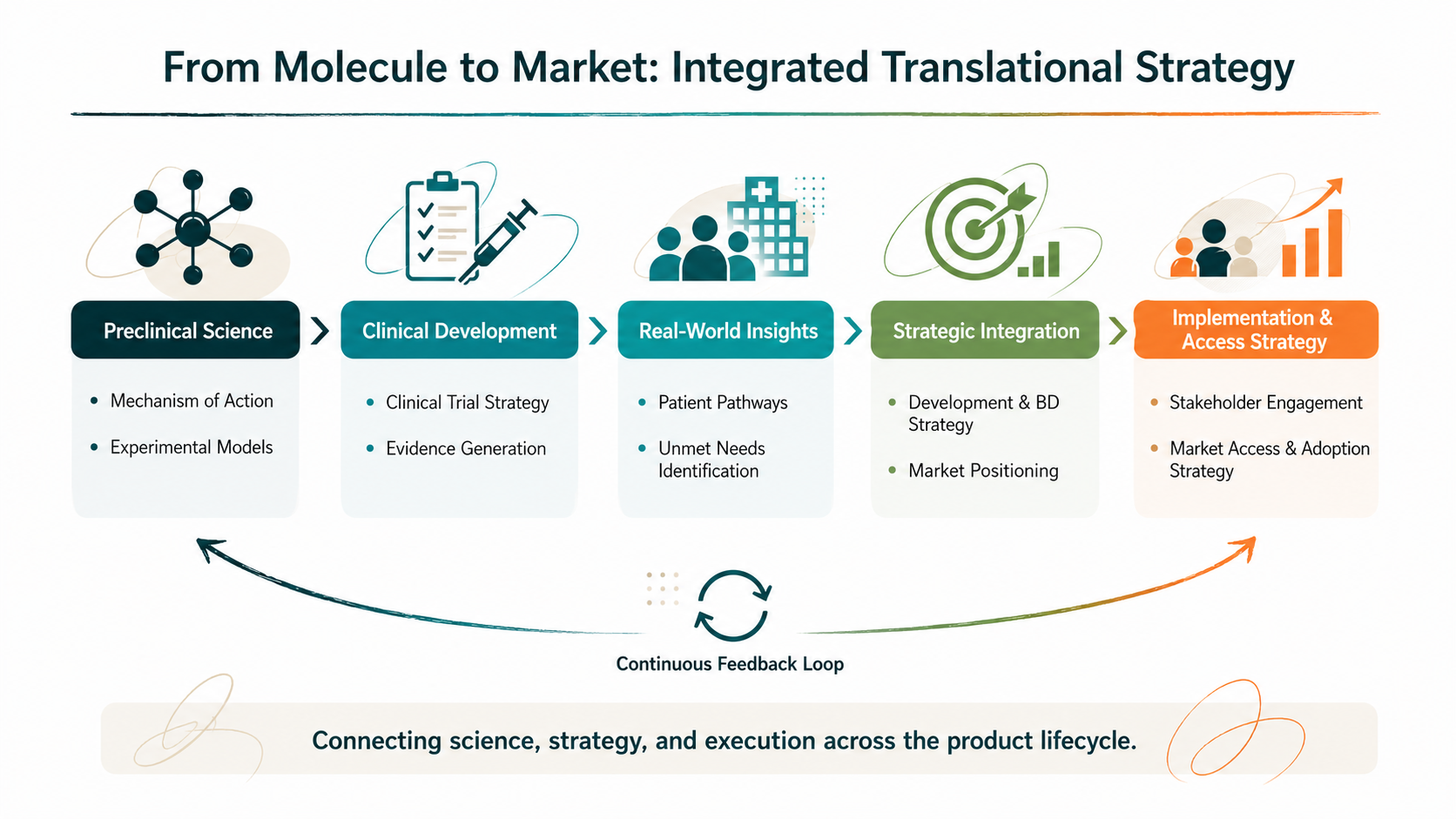
While Pulsus Pharma Consultancy focuses on evidence generation, strategy, and implementation across the clinical and post-approval lifecycle, Pulsus Development operates upstream.
It addresses a different question:

How do we maximise the value of this asset?

Should this asset progress - and if so, how?
Pulsus Development enables earlier engagement in the product lifecycle, particularly in:
By aligning early scientific insight with future clinical and commercial realities, it reduces downstream risk and supports more confident investment decisions.