Our approach is grounded in end-to-end translational science:
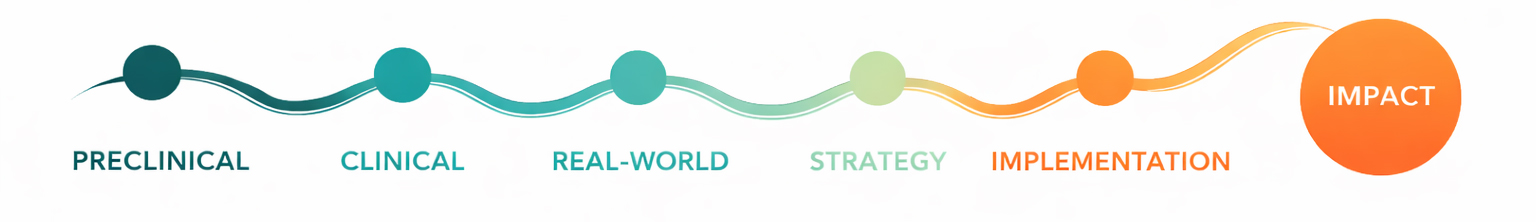
We integrate preclinical evidence, clinical development, and real-world practice to ensure that scientific insights are interpreted in context and translated into actionable strategy and effective implementation.
We operate in areas where complexity demands integration of preclinical science, clinical development, and real-world practice.
Our core focus includes neuroscience and psychiatry, oncology, and rare diseases, with additional experience in areas such as women’s health and ophthalmology.
We support clients across multiple geographies, combining global strategic insight with a deep understanding of local market dynamics.
Our experience spans:
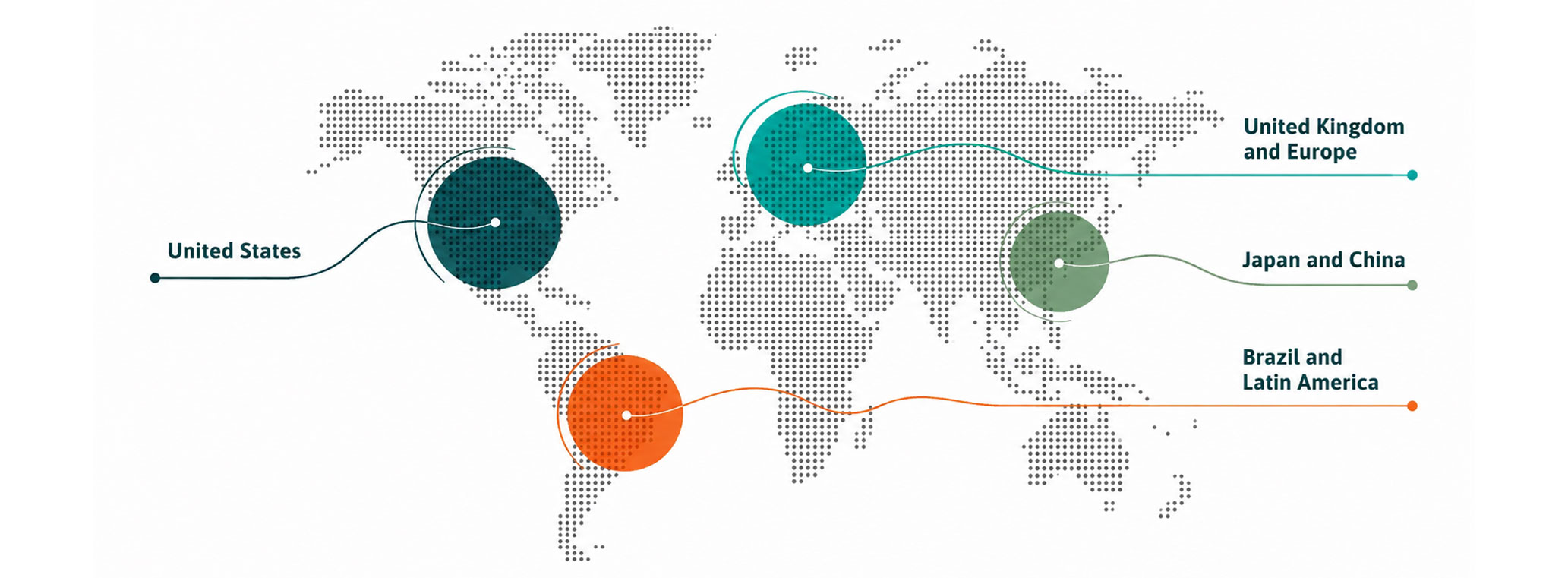
We bring global standards to local decision-making and local insight to global strategy.
A core Pulsus capability is the Embedded Implementation Lead, a role designed to ensure that strategy is effectively translated into real-world execution.
Working within client teams, it aligns functions, engages stakeholders, and adapts plans to local contexts, accelerating implementation and maximising impact.
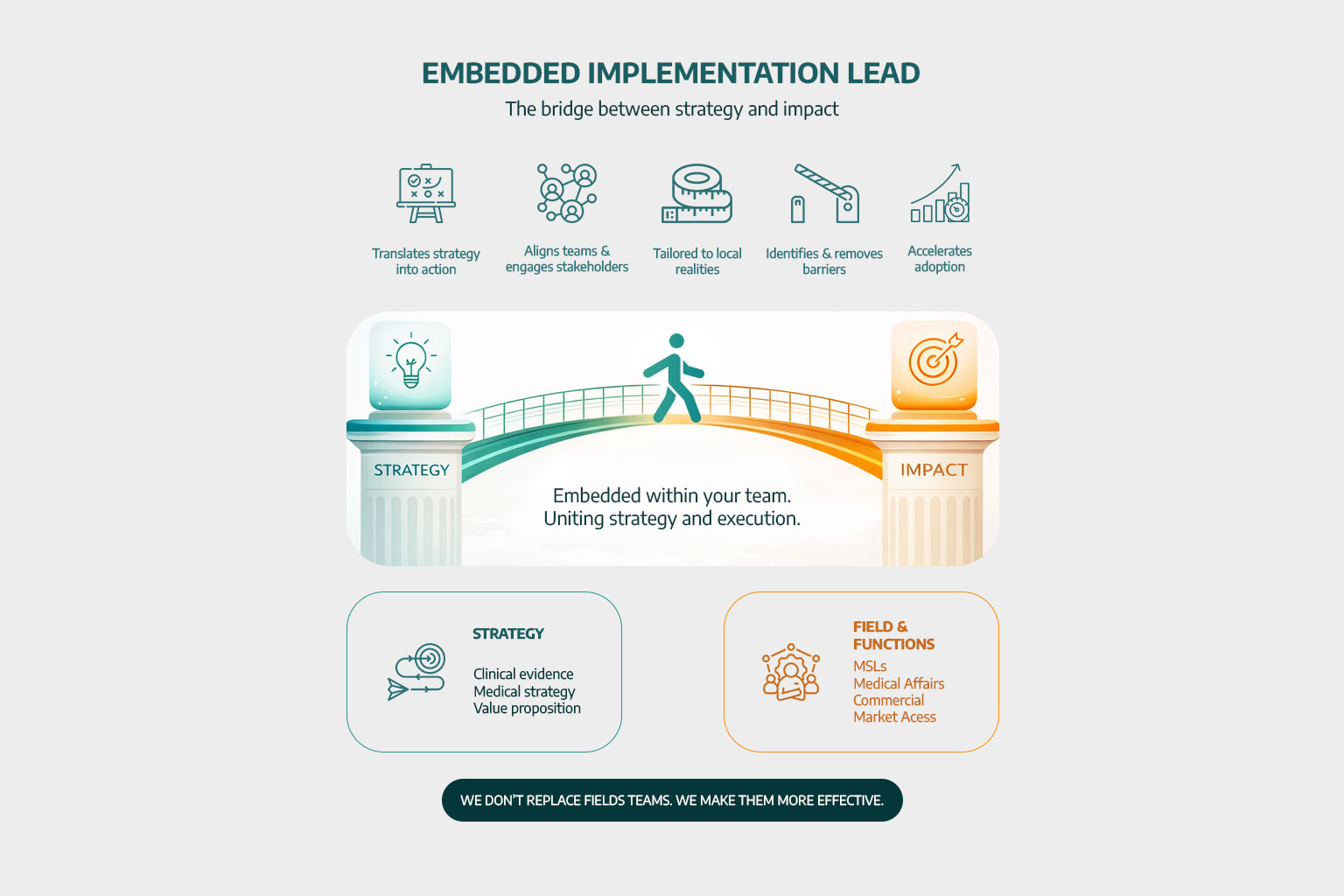
As a boutique consultancy, Pulsus offers highly tailored, senior-led support adapted to each client’s needs. We combine strategic insight with hands-on execution, working flexibly across projects rather than applying standardised approaches.
Trusted by leading pharmaceutical, biotech, and diagnostics companies across Europe, Asia, the United States, and Latin America.
Our collaborations combine global strategic insight with deep understanding of local market dynamics. We partner closely with client teams to address complex scientific, clinical, and commercial challenges, from early-stage development to real-world implementation.
Our work is typically embedded within cross-functional teams, contributing to high-impact programmes across medical affairs, clinical development, regulatory strategy, commercial and market access.














Selected collaborations include global pharmaceutical companies, biotech innovators, and diagnostics organisations.
Senior-led expertise integrating preclinical science, clinical development, and real-world implementation
Founder & CEO
Mauricio Silva de Lima is the Founder of Pulsus Pharma Consultancy and leads its strategic direction across global markets. He has authored over 100 peer-reviewed publications and scientific presentations across key therapeutic areas.
With extensive experience in clinical development, medical affairs, regulatory strategy, and evidence generation – including scientific publications – he specialises in linking preclinical insights with clinical and real-world outcomes.
His work is grounded in rigorous, evidence-based methodologies, such as systematic reviews and meta-analyses, combined with implementation science to ensure that scientific innovation translates into meaningful clinical and commercial impact.


Preclinical and Translational Research Lead
Pedro Antonio Schmidt do Prado Lima is the Preclinical and Translational Research Lead at Pulsus Development, specialising in early-stage drug development and the critical evaluation of preclinical evidence.
With extensive experience supporting pharmaceutical programmes—particularly in collaboration with industry partners in Brazil—his work focuses on mechanism-of-action assessment, experimental models, and the rigorous appraisal of early scientific data to inform downstream development decisions.
In addition to his research background, Pedro brings significant clinical experience, enabling him to assess the clinical relevance of preclinical findings and anticipate challenges in their application. His work ensures that early scientific insights are grounded in both biological plausibility and clinical reality.

AI Medical Affairs & Clinical Development
Dr. Alf Bernhardt is a Senior Advisor to Pulsus Pharma Consultancy, bringing over 25 years of experience in oncology, hematology, and cell therapy. He advises pharmaceutical and biotech companies on medical affairs strategy, clinical development, evidence generation, and product launch execution.
He has held senior leadership roles at Roche, Seagen (Pfizer), and Takeda, leading global medical affairs across the product lifecycle, including major oncology launches and the build-out of European medical organisations.
His expertise spans medical affairs strategy, clinical development (Phase I–IV), KOL engagement, evidence generation, regulatory strategy, and product launch execution, with a focus on cell and gene therapy and the application of AI in medicine. His work supports the effective translation of scientific innovation into clinical adoption and real-world impact.
He is the founder of Alf Bernhardt Medical Consulting (Switzerland; abmedicalconsulting.ch) and serves as an independent consultant to NemetzGroup LLC (Boston).

Junior Consultant – Real-World Evidence & Data Analytics
Henrique Andrea de Lima is a Biomedical Sciences undergraduate at City St George’s, University of London. Fluent in English and Portuguese, this dual Brazilian-Italian citizen has lived across California, Switzerland, and the UK, bringing a global perspective to medical affairs.
Having interned within the Real-World Evidence (RWE) team at Roche Brazil, Henrique utilizes advanced statistical models and data manipulation tools (R, SQL, DBeaver) to drive evidence-based decision-making. Aligning perfectly with Pulsus’s focus on bottom-up evidence generation, he pairs these technical capabilities with a robust clinical research background, having co-authored psychiatric research on ADHD and routinely participated in conducting rigorous systematic reviews for evidence synthesis.
At Pulsus, strategy is developed through close integration of preclinical science, clinical development, and real-world practice. This ensures that early evidence is interpreted in a clinically meaningful context, supporting regulatory decision-making, market access, and effective implementation.